中央研究院 生物化學研究所
中央研究院 生物化學研究所
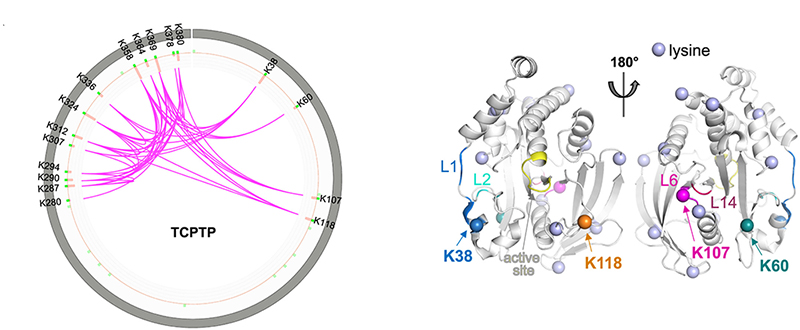
Since the discovery of protein tyrosine phosphorylation as one of the critical post-translational modifications, it has been well known that the activity of protein tyrosine kinases (PTKs) is tightly regulated. On the other hand, protein tyrosine phosphatases (PTPs) are often regarded to act constitutively active, but recently we and others have shown that many PTPs are expressed in an inactive form due to allosteric inhibition by their unique structural features. Furthermore, their cellular activity is highly regulated in a spatiotemporal manner. In general, PTPs share a conserved catalytic domain comprising about 280 residues that is flanked by either an N-terminal or a C-terminal non-catalytic segment, which differs significantly in size and structure from each other and is known to regulate specific PTP's catalytic activity. The well-characterized non-catalytic segments can be globular or intrinsically disordered. In this work, we have focused on the T-Cell Protein Tyrosine Phosphatase (TCPTP/PTPN2) and demonstrated how the hybrid biophysical-biochemical methods can be applied to unravel the underlying mechanism through which TCPTP's catalytic activity is regulated by the non-catalytic C-terminal segment. Our analysis showed that TCPTP is auto-inhibited by its intrinsically disordered tail and trans-activated by Integrin alpha-1's cytosolic region.