中央研究院 生物化學研究所
中央研究院 生物化學研究所
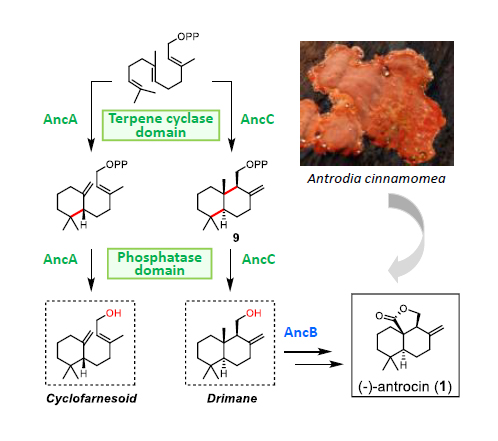
(-)-Antrocin (1), produced by the medicinal mushroom, Antrodia cinnamomea, is a potent anti-proliferative compound. The biosynthetic gene cluster of 1 is identified and the pathway is characterized by heterologous expression. We characterized a haloacid dehalogenase-like terpene cyclase AncC that biosynthesizes the drimane-type sesquiterpene (+)-albicanol (2) from farnesyl pyrophosphate (FPP). Biochemical characterization of AncC, including kinetic studies and mutagenesis, demonstrated the functions of two domains - a terpene cyclase (TC) and a pyrophosphatase (PPase). The TC domain first cyclizes FPP to albicanoyl pyrophosphate and the PPase domain then removes the pyrophosphate forming 2. Intriguingly, AncA (94% sequence identity to AncC), in the same gene cluster, converts FPP to (R)-trans-γ-monocyclofarnesol instead of 2. Notably, Y283/F375 in the TC domain of AncA serve as a gatekeeper in controlling the formation of cyclofarnesoid rather than drimane-type scaffold.