中央研究院 生物化學研究所
中央研究院 生物化學研究所
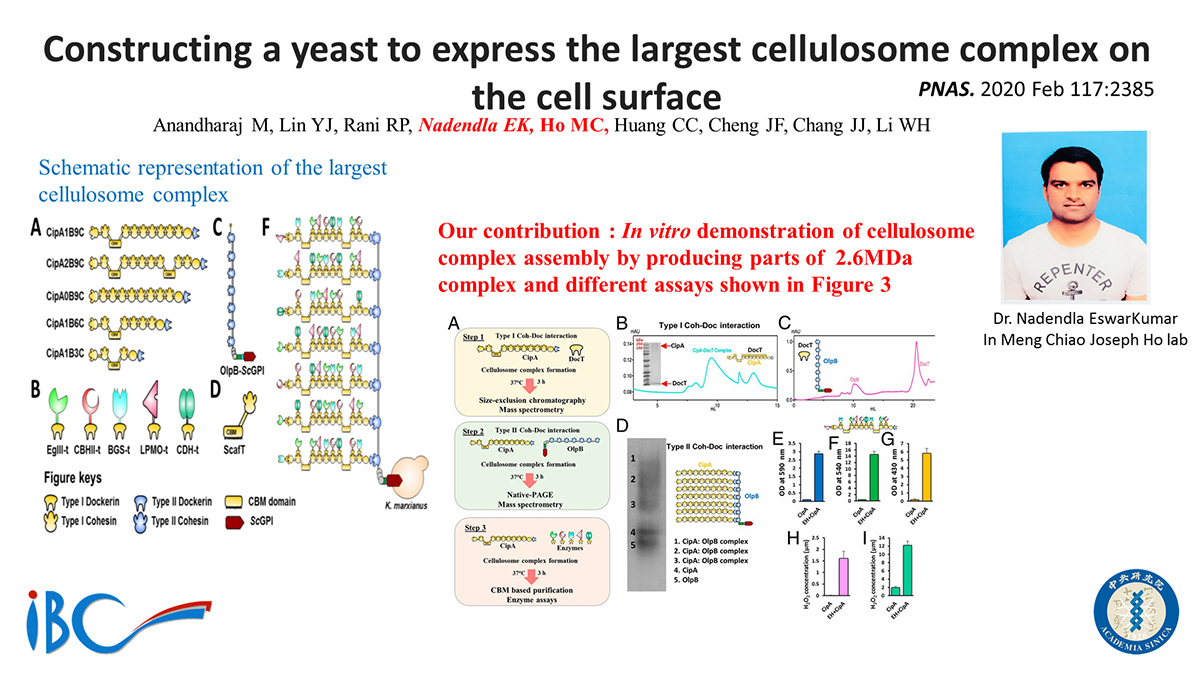
Cellulosomes, which are multienzyme complexes from anaerobic bacteria, are considered nature's finest cellulolytic machinery. Thus, constructing a cellulosome in an industrial yeast has long been a goal pursued by scientists. However, it remains highly challenging due to the size and complexity of cellulosomal genes. Here, we overcame the difficulties by synthesizing the Clostridium thermocellum scaffoldin gene (CipA) and the anchoring protein gene (OlpB) using advanced synthetic biology techniques. The engineered Kluyveromyces marxianus, a probiotic yeast, secreted a mixture of dockerin-fused fungal cellulases, including an endoglucanase (TrEgIII), exoglucanase (CBHII), β-glucosidase (NpaBGS), and cellulase boosters (TaLPMO and MtCDH). The confocal microscopy results confirmed the cell-surface display of OlpB-ScGPI and fluorescence-activated cell sorting analysis results revealed that almost 81% of yeast cells displayed OlpB-ScGPI. We have also demonstrated the cellulosome complex formation using purified and crude cellulosomal proteins. Native polyacrylamide gel electrophoresis and mass spectrometric analysis further confirmed the cellulosome complex formation. Our engineered cellulosome can accommodate up to 63 enzymes, whereas the largest engineered cellulosome reported thus far could accommodate only 12 enzymes and was expressed by a plasmid instead of chromosomal integration. Interestingly, CipA 2B9C (with two cellulose binding modules, CBM) released significantly higher quantities of reducing sugars compared with other CipA variants, thus confirming the importance of cohesin numbers and CBM domain on cellulosome complex. The engineered yeast host efficiently degraded cellulosic substrates and released 3.09 g/L and 8.61 g/L of ethanol from avicel and phosphoric acid-swollen cellulose, respectively, which is higher than any previously constructed yeast cellulosome.