中央研究院 生物化學研究所
中央研究院 生物化學研究所
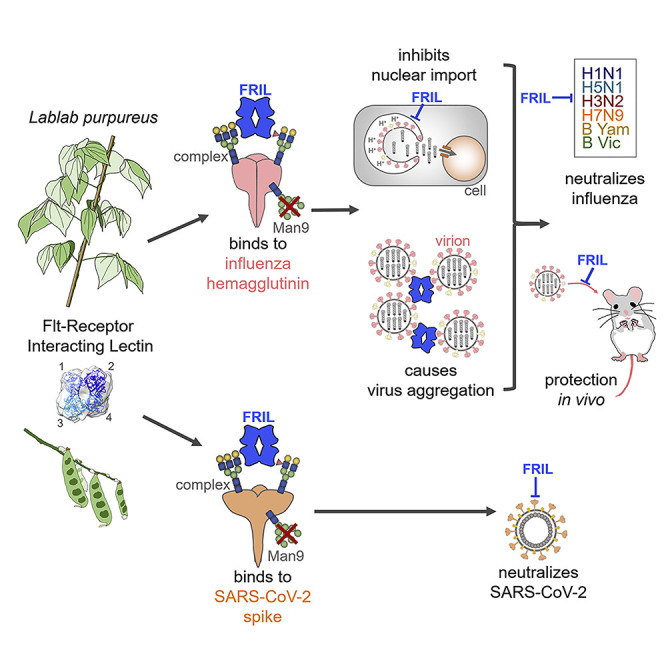
The influenza virus hemagglutinin (HA) and coronavirus spike (S) protein mediate virus entry. HA and S proteins are heavily glycosylated, making them potential targets for carbohydrate binding agents such as lectins. Here, we show that the lectin FRIL, isolated from hyacinth beans (Lablab purpureus), has anti-influenza and anti-SARS-CoV-2 activity. FRIL can neutralize 11 representative human and avian influenza strains at low nanomolar concentrations, and intranasal administration of FRIL is protective against lethal H1N1 infection in mice. FRIL binds preferentially to complex-type N-glycans and neutralizes viruses that possess complex-type N-glycans on their envelopes. As a homotetramer, FRIL is capable of aggregating influenza particles through multivalent binding and trapping influenza virions in cytoplasmic late endosomes, preventing their nuclear entry. Remarkably, FRIL also effectively neutralizes SARS-CoV-2, preventing viral protein production and cytopathic effect in host cells. These findings suggest a potential application of FRIL for the prevention and/or treatment of influenza and COVID-19.