中央研究院 生物化學研究所
中央研究院 生物化學研究所
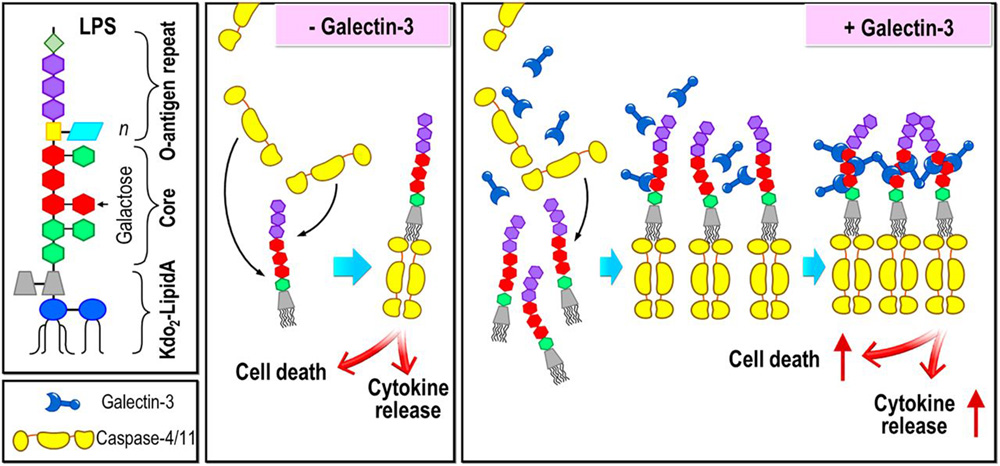
Cytosolic lipopolysaccharides (LPSs) bind directly to caspase-4/5/11 through their lipid A moiety, inducing inflammatory caspase oligomerization and activation, which is identified as the noncanonical inflammasome pathway. Galectins, β-galactoside-binding proteins, bind to various gram-negative bacterial LPS, which display β-galactoside-containing polysaccharide chains. Galectins are mainly present intracellularly, but their interactions with cytosolic microbial glycans have not been investigated. We report that in cell-free systems, galectin-3 augments the LPS-induced assembly of caspase-4/11 oligomers, leading to increased caspase-4/11 activation. Its carboxyl-terminal carbohydrate-recognition domain is essential for this effect, and its N-terminal domain, which contributes to the self-association property of the protein, is also critical, suggesting that this promoting effect is dependent on the functional multivalency of galectin-3. Moreover, galectin-3 enhances intracellular LPS-induced caspase-4/11 oligomerization and activation, as well as gasdermin D cleavage in human embryonic kidney (HEK) 293T cells, and it additionally promotes interleukin-1β production and pyroptotic death in macrophages. Galectin-3 also promotes caspase-11 activation and gasdermin D cleavage in macrophages treated with outer membrane vesicles, which are known to be taken up by cells and release LPSs into the cytosol. Coimmunoprecipitation confirmed that galectin-3 associates with caspase-11 after intracellular delivery of LPSs. Immunofluorescence staining revealed colocalization of LPSs, galectin-3, and caspase-11 independent of host N-glycans. Thus, we conclude that galectin-3 amplifies caspase-4/11 oligomerization and activation through LPS glycan binding, resulting in more intense pyroptosis-a critical mechanism of host resistance against bacterial infection that may provide opportunities for new therapeutic interventions.