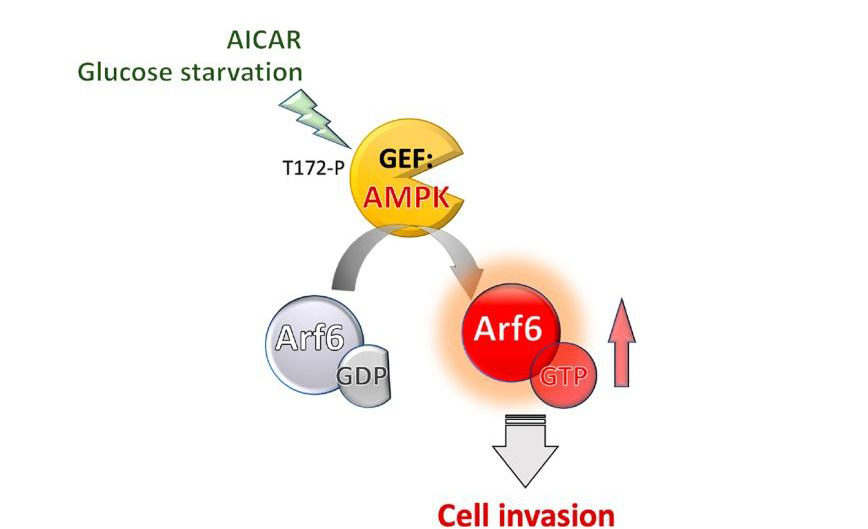
AMP-activated protein kinase (AMPK) is a crucial cellular nutrient and energy sensor that maintains energy homeostasis. AMPK also governs cancer cell invasion and migration by regulating gene expression and activating multiple cellular signaling pathways. ADP-ribosylation factor 6 (Arf6) can be activated via nucleotide exchange by guanine nucleotide exchange factors (GEFs), and its activation also regulates tumor invasion and migration. By studying GEF-mediated Arf6 activation, we elucidated that AMPK functions as a noncanonical GEF for Arf6 in a kinase-independent manner. Moreover, by examining the physiological role of the AMPK-Arf6 axis, we determined that AMPK activates Arf6 upon glucose starvation and 5-aminoimidazole-4-carboxamide-1-β-D-ribofuranoside (AICAR) treatment. We further identified the binding motif in the C-terminal regulatory domain of AMPK that is responsible for promoting Arf6 activation and thus inducing cell migration and invasion. These findings reveal a noncanonical role of AMPK in which its C-terminal regulatory domain serves as a GEF for Arf6 during energy deprivation.

 Institute of Biological Chemistry, Academia Sinica
Institute of Biological Chemistry, Academia Sinica